Pharma serialisation made simple
Easy-to-implement serialisation software in compliance with the EMVO requirements.
Robust serialisation and reporting software
Trax2D is designed to integrate into your existing setup, with zero downtime to your operations. You also get a single point of contact for technical support.
Complete serialisation solution
The Trax2D wizard offers a step-by-step process to create serial numbers, package identification, and labels for pharma stock. An ideal solution for any Parallel Importer.
Rapid and affordable compliance
Trax2D helps you to achieve compliance with FMD & EMVO regulations. The automated elements of the software make the process of regulatory compliance fast and efficient. Trax2D integrates with the existing infrastructure with zero operations downtime.
Granular permissions
The application can be customised to match the company’s existing processes with user role permissions to reflect user access. Single sign-on support allows the company to configure SSO with Active Directory, Office 365, Google and other SAML 2.0 compliant systems.
Enhance package design
Trax2D can directly feed serialised data to popular label design packages such as Bartender, NiceLabel, ZebraDesigner, and other custom applications.
Everything you need to easily create serial numbers and reach EMVO compliance in one affordable software package.
1
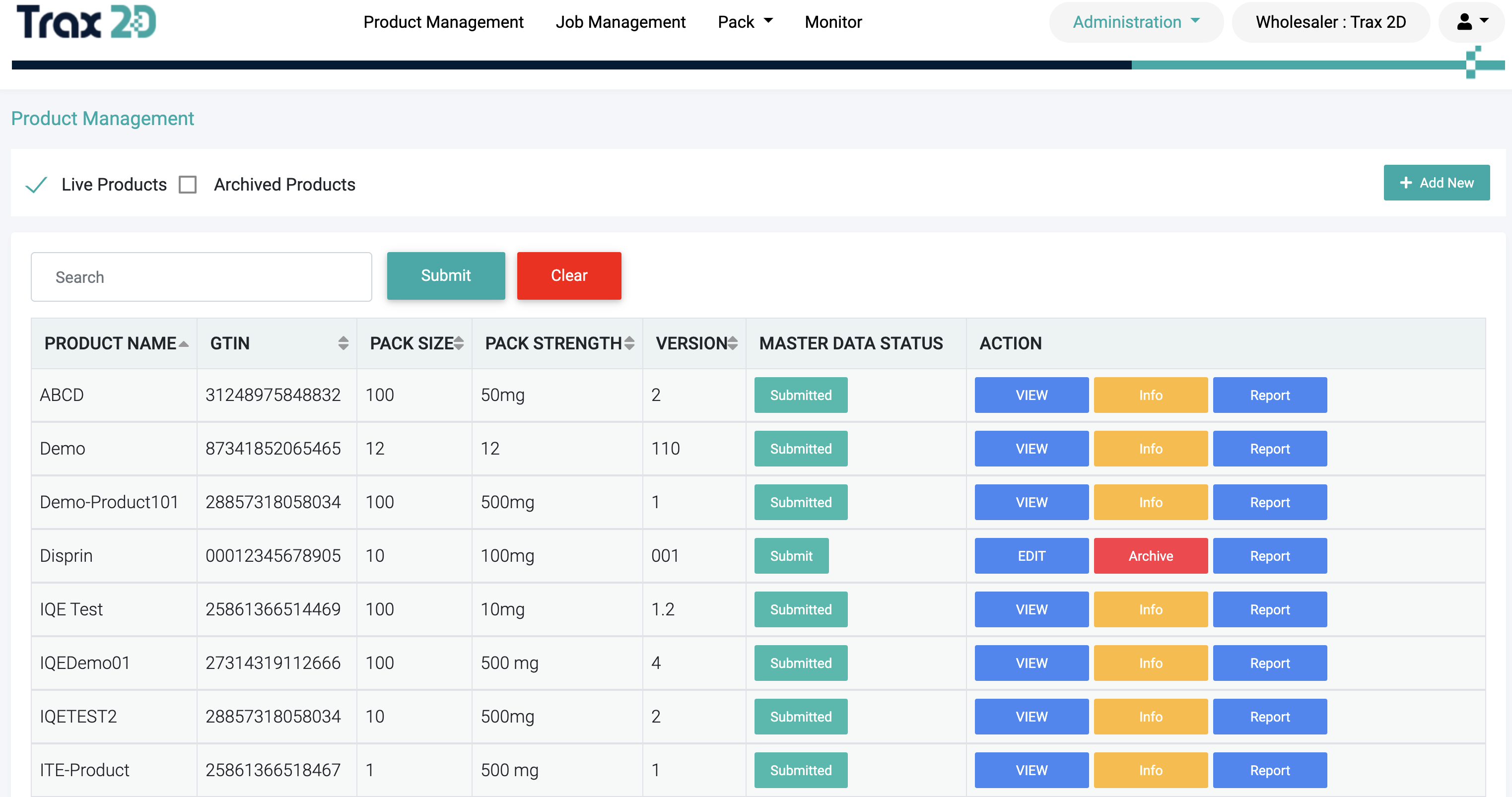
Product Master Data
Trax2D allows Manage all the records, including GTIN management, version control,
internal approval processes and EMVO upload.
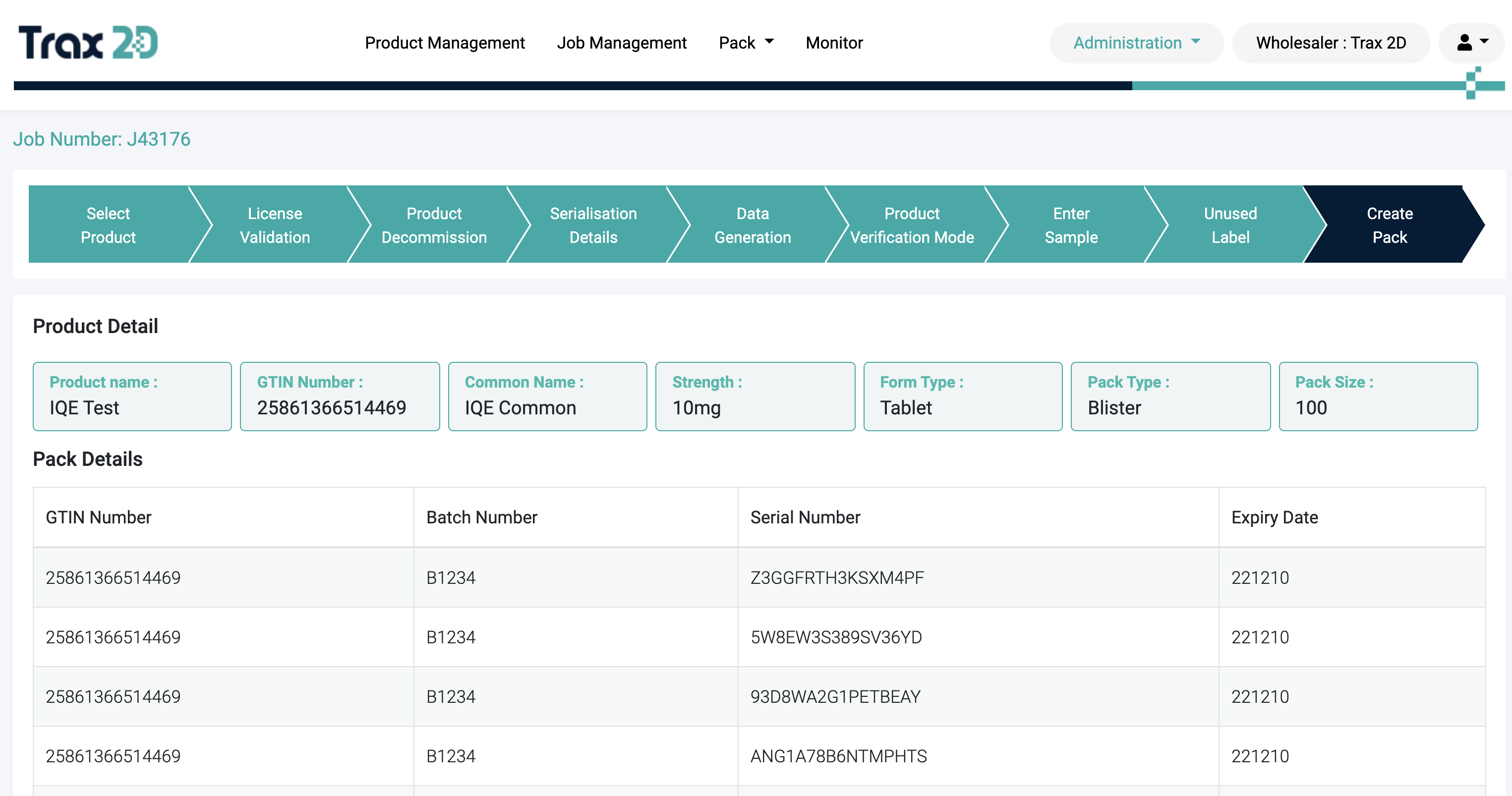
2
Serialisation
Create and upload pack data to the EMVO with a wizard-driven approach that matches warehouse operational steps. The
wizard can be tailored to integrate with current systems and with the added benefit of granular permissions.
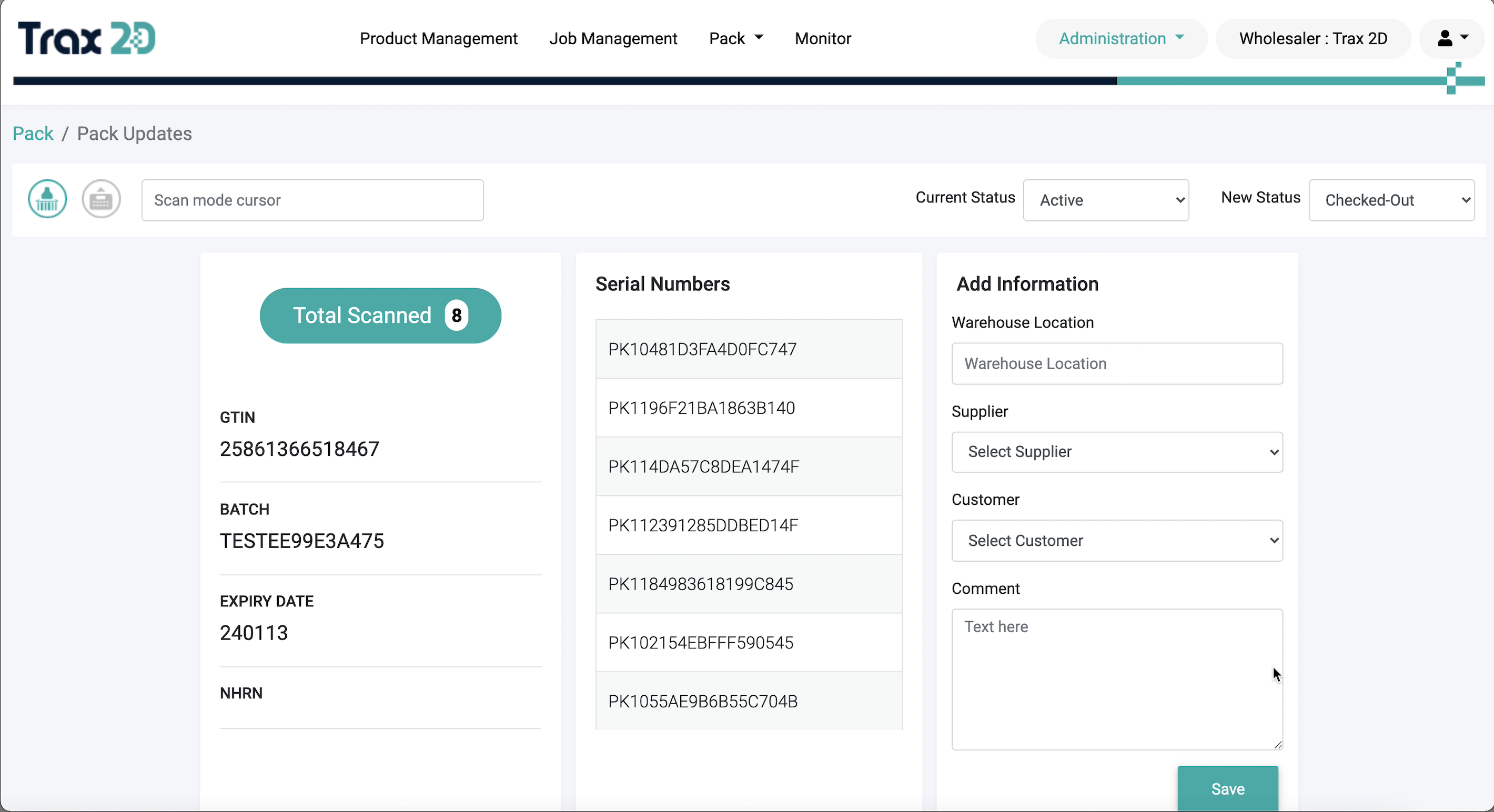
3
Pack Status
Check and change the state of packs as per requirements. With Trax2D, users can process aggregation codes
generated from FMVerify to speed up the decommissioning process.
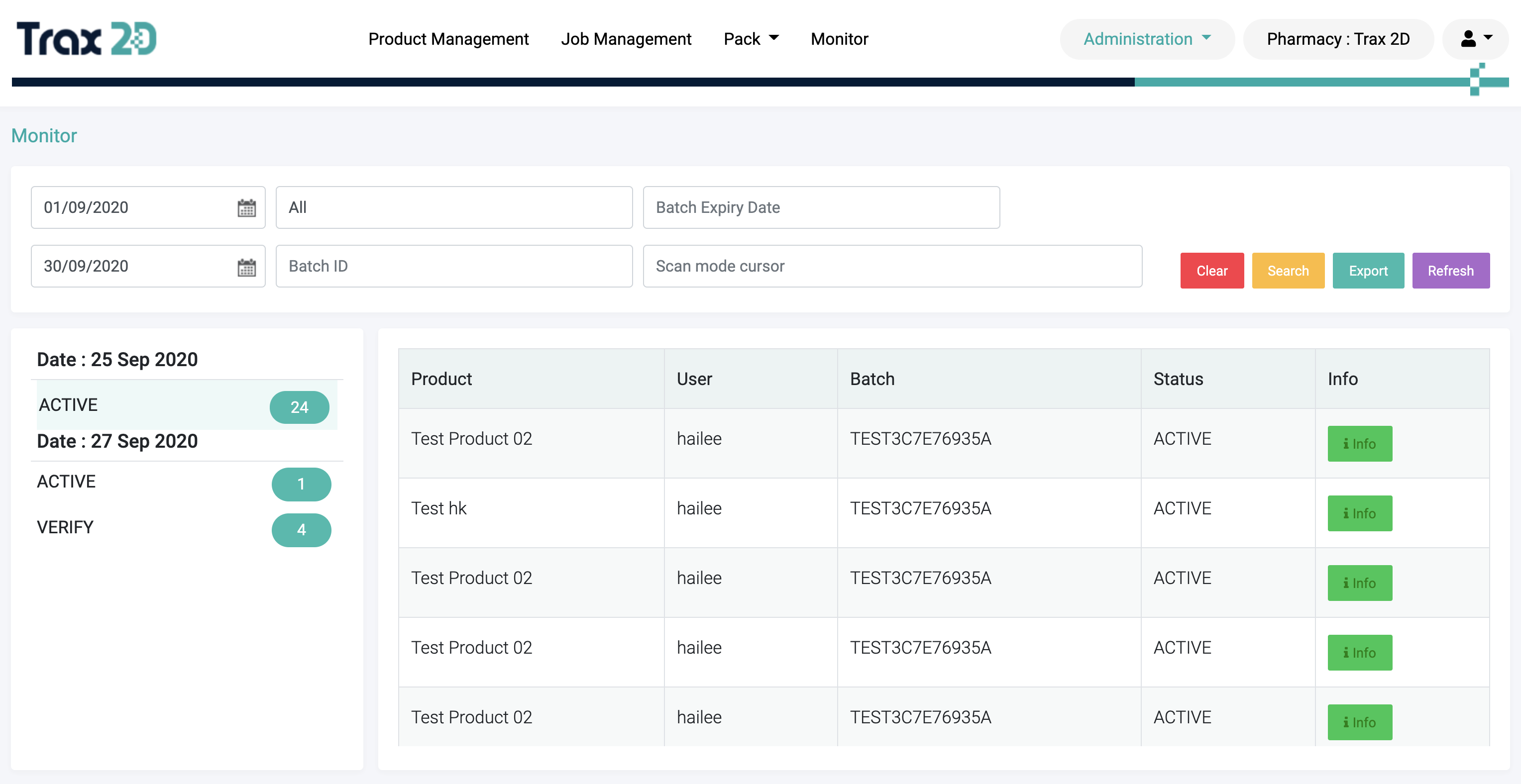
4
Monitor Reports
Monitor scanned data & automatically produce audit, batch, and
expiry reports. Export data available in Excel format.

